
NAMs: a paradigm change in biomedical research and toxicology
A CONTRIBUTION FROM PRO ANIMA SCIENTIFIC BOARD
Historically, animal models have been a central pillar of biomedical, pharmacological and toxicological research. While they have enabled significant advances, their limitations are now well documented, particularly in terms of their transferability to humans. These findings have led to the emergence of New Approach Methodologies (NAM), which bring together a set of tools and approaches based on human biology, modelling and data integration.
In line with European Directive 2010/63/EU, which encourages replacement, NAMs are gradually establishing themselves as the solutions of choice for meeting the needs of biomedical research and risk assessment. More than just alternatives, NAMs represent a paradigm shift that leverages advances in cell engineering, microfluidics, and computer modelling to better represent the complexity of the human body.
This article aims to present the diversity of these new models, their applications, their contributions and their limitations, in order to fuel societal debate and inform public decision-making.
What are New Approach Methodologies?
The acronym NAM is a generic term that encompasses a wide range of methods, mainly non-animal, used to explore human biology and generate data on the biological and toxic effects of substances.
In simple terms, NAMs seek to answer questions such as: Is a substance effective? Is it toxic? At what dose? Through what biological mechanisms?, using scientific strategies that differ from those used in animal models. NAMs are not strictly defined in most regulations. Instead, they are referred to using terms such as “alternative or substitute” or “non-animal” methods. Nevertheless, the term NAM is gradually becoming established to encompass these innovative approaches in scientific, regulatory and industrial circles.
The main categories of NAMs
In vitro methods: Cell/tissue culture models
In vitro methods are a long-standing and central pillar of NAMs. They involve culturing cells or tissues, often of human origin, in the laboratory in order to observe their responses to substances or physiological or pathological stimuli.
Models are simple when they comprise a single cell type. In recent years, more complex systems have been developed with different types of cells cultured in 3D: organoids, reconstructed tissues, tissue sections. Tumoroids, for example, are structures formed from cells taken from patients’ tumours.
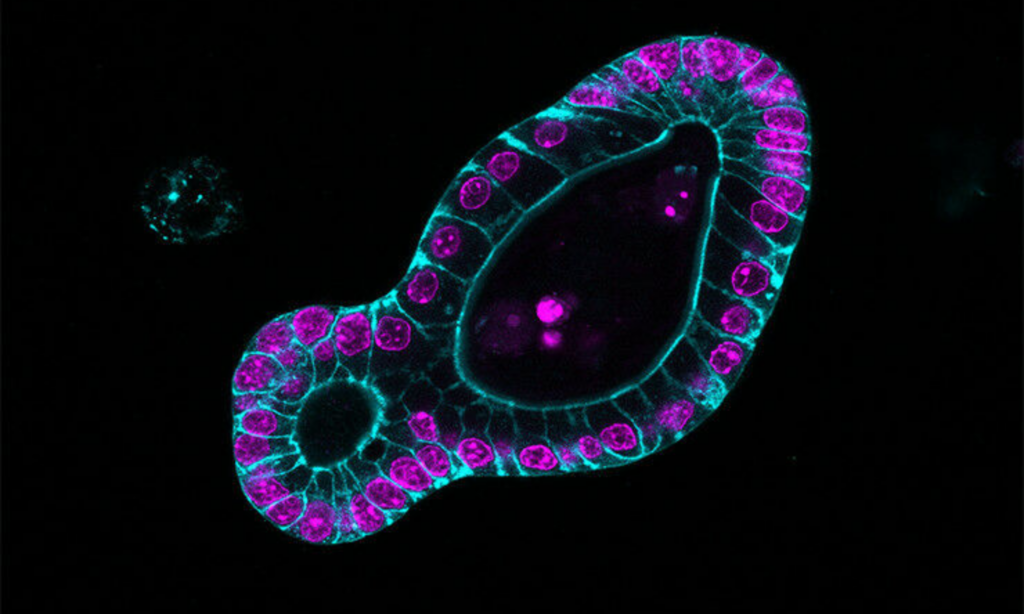
Intestine organoid. Image credits: Prisca Liberali/FMI
The most advanced models, such as organs-on-chip (OOC), use fluid circulation in microchannels (known as microfluidics) to precisely control the microenvironment and thus reproduce the interactions between the different cell types that make up an organ, as well as with the dynamic environment in which they live. These microphysiological systems (MPS) involve modulating mechanical and environmental stresses to more accurately reproduce certain aspects of tissue architecture, cellular diversity and interactions specific to human organs.

Organ-on-chip. Image credits: NETRI
Some examples of in vitro models
- Reconstructed human skin or eye models for testing irritation;
- More advanced three-dimensional systems, organoids or organ-on-a-chip devices, capable of reproducing certain key functions of human organs;
- Microfluidics coupled with organ-on-a-chip technology to better understand the process of metastasis development, for high-throughput screening of active molecules, or to test the efficacy of treatments.
In silico approaches: mathematical modelling, bioinformatics and AI
In silico methods are based on computer models that predict the behaviour of substances in biological systems and their interactions with certain targets such as nuclear or membrane receptors, proteins that modify the activity and/or function of cells in the body. They use existing databases, mathematical models and artificial intelligence tools.
Among the most common examples in toxicology:
- Read-across approaches, which assume that two chemically similar substances will have similar effects [1];
- SAR (structure – activity relationship) models capable of linking the chemical structure of a molecule to its biological or toxic effects, in some cases quantitatively (QSAR) [2];
- PBPK (pharmacokinetics based on physiology) models, which estimate the absorption, distribution, metabolism and elimination of a substance in the human body.
These methods and others (such as 3D modelling) are particularly useful for the initial screening of large libraries of substances, to limit or even replace the usual experimental testing. They also provide, in record time, information on the behaviour of these substances in the body and the targets they are able to reach.
A rapidly growing concept originating in industry, the digital twin in healthcare is a dynamic model of a patient, organ or physiological process, built from medical, biological, environmental and behavioural data. It can be used to simulate the progression of a disease or the potential effect of a treatment with a view to providing more personalised medicine.

Focus: Mathematics for numerical modelling
Mathematics plays a central role in in silico approaches, providing the formal language needed to represent biological phenomena and predict system behaviour. For example, differential equations describe the dynamics within PBPK models, while statistical and multivariate methods enable the construction of (Q)SAR models and the quantification of uncertainties. Optimisation algorithms enable parameter adjustment, sensitivity analysis and scenario exploration. Mathematics also forms the basis of AI-enhanced NAMs, which are capable of transforming biological complexity into computable predictive models, thereby driving safer and more efficient research.
In chemico methods: chemistry as a biological indicator
In chemico approaches focus on the key chemical reactions involved in toxicity mechanisms, particularly the interaction of substances with proteins (which cause allergic reactions) or with DNA (which causes harmful genetic mutations). They are widely used to measure the effect of substances on skin proteins, which can cause skin sensitisation. Today, the combination of in vitro and cell-based tests has replaced most animal testing for skin sensitisation, although some immunological tests are still performed in mice. In the field of drug development, in vitro tests are used to assess potential side effects or possible interactions with other drugs.

Integrated approaches: combining evidence
One of the major developments in NAMs is the shift from isolated tests to integrated assessment frameworks, combining different data sources to support regulatory decisions. Key concepts include: Adverse Outcome Pathways (AOPs), which describe the chain of events linking an initial molecular interaction to an observable adverse effect; Integrated Approaches to Testing and Assessment (IATA), which rely on combining in silico, in chemico and in vitro data; and Next Generation Risk Assessment (NGRA) approaches, which integrate hazard, exposure and mechanism of action data to assess real risks to humans.
In addition, there are “omic” analyses (transcriptomics, proteomics, lipidomics, metabolomics) to measure molecular and biochemical responses in cells, tissues and biological fluids. In toxicology, these approaches are used to characterise and quantify the effects of exposure to chemicals or drugs, contributing to a more mechanistic and predictive assessment.

Focus: Myths and misconceptions about NAMs
Some common misconceptions about NAMs:
- “NAMs imply the immediate or total end of all animal suffering.” This perception is based on a confusion of terminology. NAMs can be defined as Non-Animal Methods, but they also refer to New Methodological Approaches. Although they are alternatives to animal experimentation, in vitro NAMs may use products of animal origin (serums or matrices).
- “NAMs are already fully accepted by regulatory authorities.” While agencies are beginning to incorporate NAMs into their recommendations and have accepted some of them, traditional toxicological testing on animals remains widely required.
- “NAMs are already sufficient to replace all animal testing as a single or universal solution.” NAMs are based on a reasoned combination of models and data, designed to answer specific questions. Their objective is not to “do without animals at all costs”, but to produce data that is more relevant to humans, within a rigorous and transparent scientific framework.
In which areas are NAMs already being used?
Safety assessment of substances
NAMs are incorporated into regulatory assessment in several sectors. Since 2013, animal testing has been prohibited in the EU for cosmetic products and their ingredients when validated alternative methods exist, such as reconstructed human skin models.
In chemical regulations, particularly REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals), companies must avoid animal testing. NAMs (in vitro, in chemico and in silico) are used via weight-of-evidence approaches; animal testing should only be used as a last resort.
In the food sector, risk assessments (additives, contaminants, contact materials, etc.) are still largely based on animal data, but European authorities are aiming to increase the proportion of data derived from NAMs. Similarly, regulations on biocides and plant protection products (pesticides) promote the reduction of animal testing.
NAMs are also transforming the evaluation of medical devices. Agencies are encouraging their use (particularly in the United States and Japan). Despite these advances, obstacles remain (see next chapter).
NAMs in biomedical research
In biomedical research, NAMs, in vitro and in silico, offer original perspectives not only for better understanding diseases and discovering new targets (biomarkers), but also for developing and testing new therapeutic strategies. Today, approximately 90% of drug candidates validated in preclinical animal tests fail in clinical trials [1,2]; contributing to a long and costly development process. NAMs, which are more predictive, faster and less costly, have the potential to improve the R&D cycle: understanding mechanisms, screening and optimising therapeutic candidates, evaluating efficacy and side effects, etc.
Another perspective is to be able to offer treatments tailored to each patient or group of patients, with a view to “stratified” or “personalised” medicine. The use of organoids and patient data combined with AI is being considered, for example, for conditions such as cancer, diabetes and neurodegenerative diseases, but also for so-called vulnerable populations (pregnant women, infants, children, etc.).
In regenerative medicine, combining advanced human models (organoids, organs-on-chips) with 3D bioprinting makes it possible to study tissue repair and regeneration mechanisms with increased biological relevance.

Focus: The added value of NAMs
Beyond ethical considerations and the reduction of animal testing, NAMs offer:
- Greater relevance for humans;
- Access to subtle biological mechanisms that are difficult to observe in global animal models;
- The ability to produce data earlier in the R&D cycle;
- Potential gains in efficiency and cost for certain stages;
- Greater consideration for specific or vulnerable populations.
Today, NAMs appear to be indispensable for understanding:
- The danger posed by exposure to tens of thousands of chemicals whose effects we do not know and for which animal testing does not offer a satisfactory solution (cost, duration, transposition to humans);
- The issue of chemical mixtures (cocktail effects).
Limitations, challenges and conditions for success
Current scientific limitations
No model, animal or otherwise, can fully represent the complexity of a human organism. Certain issues, such as long-term systemic effects, interactions between organs, and the impact of complex exposure pathways (e.g., transgenerational alterations), remain difficult to study, even with NAMs. Some tissues, stages of development, or pathological conditions do not yet have sufficiently mature or scientifically validated models. Even though the predictability of animal models for humans does not exceed 65% and the reproducibility of trials is regularly questioned, much remains to be done to move away from animal models, particularly in the decision-making process [3].
Validation and standardisation issues
In order to be widely adopted, NAMs must demonstrate their reliability, reproducibility and biological relevance for their intended use. Scientific validation establishes this credibility, in particular through inter-laboratory comparisons and clear interpretation criteria. Where possible, standardising protocols facilitates their adoption and regulatory use. For example, in accordance with the ISO 10993 standard, authorities favour in vitro (cytotoxicity, irritation, sensitisation) and chemical characterisation coupled with in silico modelling (QSAR). The standardisation of new methods, which can be a lengthy and demanding process, is essential to build confidence among authorities, industry and the public. Harmonisation and standardisation initiatives led by the Organisation for Economic Co-operation and Development (OECD) and the International Cooperation on Alternative Test Methods (ICATM) aim to address this challenge.
Regulatory approval
NAMs can be used in innovative approaches to hazard and risk assessment. Numerous initiatives and research projects are promoting greater regulatory acceptance. While some regulations are pioneering, such as those for cosmetics, and some countries encourage their use (e.g. the FDA (Food & Drug Administration), USA) and the PMDA (Pharmaceuticals and Medical Devices Agency), Japan), the regulatory framework for NAMs is still evolving. Drug Administration, USA) and the PMDA (Pharmaceuticals and Medical Devices Agency, Japan), the acceptability of these approaches remains limited at European and global levels. Chronic and environmental effects are still largely assessed on the basis of animal studies. At the same time, mutual recognition of data from NAMs still depends on the validation and standardisation of protocols. Significant work therefore needs to be done to compare existing data in order to help regulatory bodies move towards better integration of NAMs. The International Council for Harmonisation has begun updating the appendices to its guidelines -ICH S7A and S7B- (particularly for cardiac and hepatic toxicology).
Interoperability, quality and data sharing
Interoperability, quality, and data sharing are key to the rise of NAMs. The diversity of formats, models, and platforms complicates the comparison and reuse of results. However, solutions are gradually emerging, combining common standards, harmonised metadata and FAIR principles (Findable, Accessible, Interoperable, Reusable). The development of secure infrastructures and controlled sharing spaces will improve data quality and access while protecting industrial (particularly sensitive data) and regulatory issues.
Need for new skills
NAMs can be a fundamentally different source of data from animal testing, posing a significant challenge to the current system. To ensure that we are able to understand and assimilate them now and in the future, a major effort must be made in terms of training for students, researchers and experts. Training programmes must therefore reflect the complexity and interdisciplinary requirements of NAMs.
Socio-technical barriers
The implementation of a valid and accepted method is not only a matter for science, but also for politics, law and society. Out of habit or ignorance, certain influential communities of experts tend to denigrate the usefulness of NAMs and hinder their development. The transition to these new methods requires deep reflection on the foundations of modern research. It requires collective support, bringing together researchers, competent authorities, industry and civil society.
Conclusion & Opening
The diversity of uses for NAMs in academic and industrial settings is a strength, although they are not a perfect solution for all research needs. It is therefore essential not to overpromise.
However, NAMs are establishing themselves as a major scientific and economic lever for transforming biomedical research, toxicology and risk assessment on an international scale. They therefore offer public decision-makers an opportunity to base public health policies on data that is more reliable and transferable to humans, as well as industrial policies based on cutting-edge technologies.
Their use is set to grow rapidly and the robustness of the models offered will increase, with a view to better understanding and responding to contemporary challenges in health, competitiveness and sovereignty.
However, accelerating their adoption while ensuring scientific rigour and collective confidence requires a structured approach: training and uniting stakeholders who are still too fragmented, organising shared governance of the transition, and targeted, coordinated investments that are commensurate with the challenges. The speed of progress depends on the resources allocated to validation, research, stakeholder support and infrastructure development.
Without claiming to be exhaustive on a subject as rich and promising as NAMs, this article outlines its scope, main areas of research and numerous fields of application. Other publications will follow, providing more concrete examples of how these methodologies are implemented, particularly in the area of risk assessment.
Pro Anima Scientific Advisory Board
Focus: The dynamics of tNAM: a global movement
Au-delà des enjeux éthiques et de réduction de l’expérimentation animale, les NAM offrent :
La dynamique des NAM est mondiale. Aux États-Unis, la prise de conscience de l’intérêt des NAM face aux limites des modèles animaux a conduit à une évolution réglementaire majeure. L’adoption du Food and Drug Administration (FDA) Modernization Act 2.0 puis 3.0 a supprimé l’obligation historique de recourir à l’expérimentation animale pour l’enregistrement des médicaments. La FDA a publié des feuilles de route favorisant l’intégration de données issues de modèles humains. Parallèlement, les National Institutes of Health (NIH) ont réorienté leurs financements vers des approches alternatives, tandis que l’Environmental Protection Agency (EPA) s’est engagée à éliminer progressivement les tests de toxicité sur les mammifères. Ces actions s’appuient sur de solides partenariats entre agences fédérales, institutions académiques et industrie.
L’Europe s’inscrit également dans une dynamique structurée. L’Union européenne, à travers le Joint Research Center (JRC), l’Agence européenne de la chimie et l’Agence européenne des médicaments (EMA), soutient activement la validation et l’acceptation des NAM. L’European Chemicals Agency (ECHA) et l’European Partnership for Alternative Approaches to Animal Testing (EPAA) favorisent l’harmonisation des pratiques, notamment via les approches intégrées de test et d’évaluation. Plusieurs États membres se positionnent en leaders : les Pays-Bas, avec le programme TPI (Transition Programme for Innovation without animal testing), la France, avec la structuration de la filière F3OCI (Organoïdes et Organes sur Puce) et la publication d’un Livre Blanc. Le Royaume-Uni développe sa propre stratégie nationale, soutenue par des investissements ciblés et la création de centres spécialisés.
En Asie, la dynamique s’intensifie. Le Japon intègre progressivement les NAM dans les lignes directrices de la Pharmaceuticals and Medical Devices Agency (PMDA). La Corée du Sud renforce ses capacités nationales et s’engage dans des collaborations internationales, tandis que la Chine manifeste un intérêt croissant, notamment dans le secteur cosmétique.
References
[1] EFSA Scientific Committee et al., Guidance on the use of read-across for chemical safety assessment in food and feed, EFSA Journal Vol.23 (7) 2025 doi/full/10.2903/j.efsa.2025.9586
[2] (Q)SAR Assessment Framework: Guidance for the regulatory assessment of (Quantitative) Structure Activity Relationship models and predictions, Second Edition, OECD Series on Testing and Assessment, Nov. 2024, doi.org/10.1787/bbdac345-en
[3] Sun, D, et al. (2022). Why 90% of clinical drug development fails and how to improve it? Acta Pharm Sin B., 12(7). doi.org/10.1016/j.apsb.2022.02.002
[4] Mirlohi, M.S.; Yousefi, T.; Aref, A.R.; Seyfoori, A. Integrating, New Approach Methodologies (NAMs) into Preclinical Regulatory Evaluation of Oncology Drugs. Biomimetics 2025, 10, 796. doi.org/10.3390/biomimetics10120796
[5] Sewell, F, et al. (2024). New approach methodologies (NAMs): identifying and overcoming hurdles to accelerated adoption. Toxicol Res (Camb), 13(2):tfae044. doi.org/10.1093/toxres/tfae044



