
Pro Anima missions
Our engagement
Created in 1989 under the honorary presidency of Professor Théodore Monod, the Scientific Committee Pro Anima is a council of scientists, academics and members of the medical profession, all volunteers, who work with laboratories to develop and promote research programs without experimenting on animals.
From the shortcomings and limits of the animal model, the Committee aims to promote human health safety through chemical substances that are better tested, more reliable for human health, more protective of the environment and without the use of animals.
With technological progress and the advancement of scientific knowledge, it is possible to research and test chemicals differently and better than with animal experimentation
Christiane Laupie-Koechlin
Founder — General Secretary
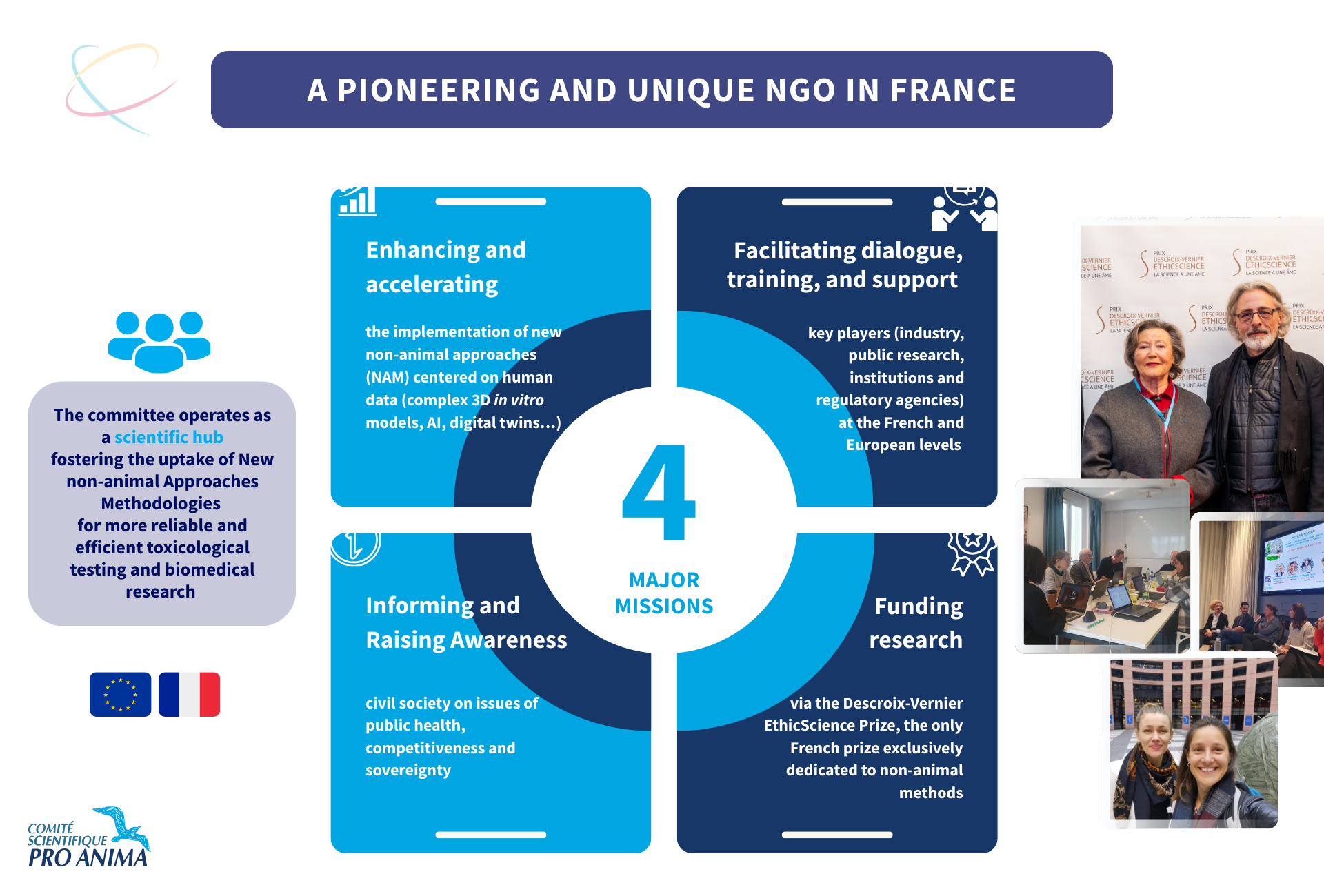
Our missions
The Committee’s main missions:
- Promote and accelerate the implementation of new non-animal approaches (NAMs) focused on human data (complex 3D in vitro models, artificial intelligence, digital twins, etc.)
- Foster dialogue, and support key stakeholders (industry, public research, institutions, and regulatory agencies)
- Raise awareness and inform civil society about the challenges of public health, innovation, competitiveness, and sovereignty
- Fund leading research through the Descroix-Vernier EthicScience Prize, the only French prize exclusively dedicated to non-animal methods
The association is free from any political, philosophical, denominational or other subjection.
The Pro Anima Scientific Committee is currently chaired by Doctor Serge Kauffer.
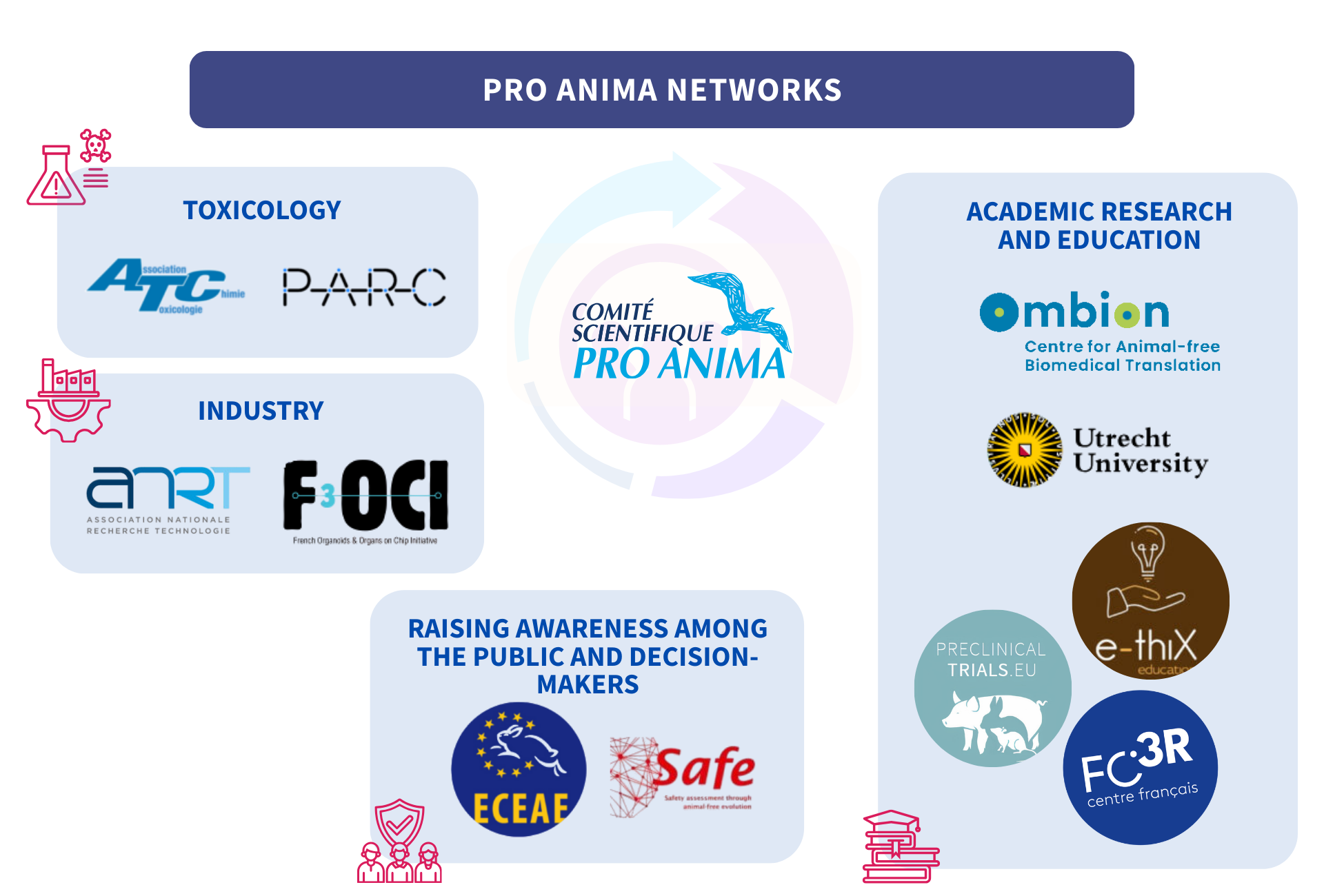
Networks
Our history in a few dates

I prefer ignorance, despite my tireless curiosity, to the acquisition of knowledge that I know was only obtained through the suffering of a living being
Pr Théodore Monod


